2San offers a rapid‚ convenient solution for detecting COVID-19‚ RSV‚ Influenza A‚ and Influenza B. This self-test utilizes a simple nasal swab‚ providing results quickly for informed healthcare decisions.
The 2San 5-in-1 test empowers individuals to proactively manage their health‚ especially during peak respiratory illness seasons‚ offering peace of mind and timely action.
Understanding the Importance of Combined Testing
Differentiating between COVID-19 and influenza (A/B) is crucial‚ as symptoms often overlap‚ leading to diagnostic uncertainty. Relying on a single test can delay appropriate treatment and public health measures. Combined testing‚ like the 2San 5-in-1 test‚ provides a comprehensive assessment of respiratory illness‚ enabling accurate diagnoses.
This approach is particularly vital during periods of co-circulation of multiple respiratory viruses. Accurate identification allows for targeted antiviral therapies‚ appropriate isolation protocols‚ and effective contact tracing. Furthermore‚ combined testing supports robust respiratory virus surveillance initiatives‚ aiding public health agencies in monitoring viral prevalence and emerging strains.
Sentinel providers contribute to this surveillance by submitting samples to PHEL‚ enhancing our understanding of the evolving respiratory landscape. Timely and accurate results from combined tests are essential for protecting individual and community health.
What is the 2San 5-in-1 Self Test?
The 2San 5-in-1 Self Test is a rapid‚ in-vitro diagnostic test designed for the simultaneous qualitative detection of SARS-CoV-2‚ Influenza A‚ Influenza B‚ and RSV antigens directly from anterior nasal swab specimens. It’s an invaluable tool for individuals experiencing respiratory symptoms‚ offering a convenient and accessible testing solution.
This test is authorized for Emergency Use Authorization (EUA)‚ intended for over-the-counter use‚ but remains for prescription use only. The test delivers results within minutes‚ aiding in quick decision-making regarding isolation and medical consultation. It differentiates between these common respiratory pathogens‚ providing clarity when symptoms overlap.
The 2San test utilizes a simple nasal swab collection method‚ making it user-friendly for self-administration‚ and is intended for use within the first five days of symptom onset for optimal accuracy.
Test Components and What They Detect
2San’s test identifies SARS-CoV-2‚ Influenza A‚ Influenza B‚ and RSV antigens from nasal swabs‚ utilizing specific antibodies to detect each virus effectively.
SARS-CoV-2 Detection
The 2San test detects the presence of the SARS-CoV-2 nucleocapsid antigen‚ a key protein component of the virus responsible for COVID-19. This detection is achieved through a specific antibody reaction on the test device‚ indicating a current infection. The test is designed to identify the virus directly from anterior nasal swab specimens.
It’s crucial to understand that this is an antigen test‚ meaning it identifies viral proteins‚ not the virus’s genetic material. Therefore‚ its accuracy is highest when testing during the initial stages of infection‚ typically within the first five days of symptom onset. Positive results suggest a current COVID-19 infection‚ while negative results may require confirmation with a molecular test‚ especially if symptoms persist.
The test’s ability to accurately detect SARS-CoV-2 is vital for timely isolation and preventing further spread within communities.
Influenza A Detection
The 2San 5-in-1 test identifies Influenza A by detecting the presence of its nucleoprotein antigen. This antigen is a specific marker for Influenza A viruses‚ allowing for accurate differentiation from other respiratory illnesses. The test utilizes antibodies that bind to this antigen‚ producing a visible result on the test device when the virus is present in the nasal sample.
Like the SARS-CoV-2 detection component‚ the accuracy of Influenza A detection is optimized when testing is performed early in the course of infection – ideally within the first five days of symptom onset. A positive result confirms an active Influenza A infection‚ while a negative result doesn’t entirely rule it out and may necessitate further testing.
Prompt identification of Influenza A is crucial for appropriate treatment and public health measures.
Influenza B Detection
The 2San 5-in-1 test also detects Influenza B‚ another common cause of seasonal flu‚ by identifying its specific nucleoprotein antigen. Similar to Influenza A detection‚ the test employs antibodies designed to bind to this unique Influenza B marker within a nasal swab sample.
A positive result indicates an active Influenza B infection‚ aiding in targeted treatment decisions. Conversely‚ a negative result doesn’t definitively exclude infection‚ particularly if testing is performed later in the illness or if viral load is low. The test’s effectiveness is maximized when used within the first five days of experiencing symptoms.
Distinguishing between Influenza A and Influenza B is important for epidemiological surveillance and understanding circulating strains.
When to Use the 2San Test
Utilize the 2San test when experiencing respiratory symptoms like fever‚ cough‚ sore throat‚ or body aches‚ especially during peak illness seasons for swift diagnosis.
Symptoms Indicating a Need for Testing
Consider utilizing the 2San test if you are experiencing symptoms consistent with respiratory infections. Common indicators include fever‚ which can range from mild to high‚ and a persistent cough‚ either dry or productive. A sore throat‚ causing discomfort when swallowing‚ is another key symptom.
Additional signs prompting testing are muscle or body aches‚ fatigue‚ and headaches. Nasal congestion or a runny nose can also be present. In some cases‚ individuals may experience gastrointestinal symptoms like nausea‚ vomiting‚ or diarrhea.
It’s crucial to test if you’ve been in close contact with someone confirmed to have COVID-19‚ Influenza A‚ or Influenza B‚ even without noticeable symptoms. Early detection allows for prompt isolation and reduces further spread within the community.
Testing Timeline: Days Post-Symptom Onset
Optimal testing with the 2San test occurs within the first five days of experiencing respiratory symptoms. Viral loads are generally highest during this period‚ maximizing the accuracy of the test results. Testing early allows for quicker diagnosis and appropriate medical intervention if needed.
While the test can still be performed beyond five days‚ the sensitivity may decrease as the viral load declines. For the most reliable results‚ especially if symptoms persist‚ consider repeat testing‚ particularly within the initial timeframe.
If initial testing is negative but symptoms worsen‚ retesting after a couple of days is recommended. Remember‚ negative results don’t entirely rule out infection‚ and clinical judgment should always guide healthcare decisions.
Preparing for the Test
Before testing‚ verify the expiration date and gather all components: test device‚ extraction buffer‚ swab‚ and instructions. Ensure a clean‚ well-lit workspace is available.
Checking the Expiration Date
Crucially‚ always inspect the test kit’s packaging for the expiration date before commencing the testing procedure. Utilizing an expired test can yield inaccurate results‚ potentially leading to misinformed decisions regarding your health and the health of others. The expiration date is prominently displayed on the outer box and often on individual test device pouches.
Do not use the test if it has passed the expiration date. Expired reagents may degrade‚ compromising the test’s ability to accurately detect the presence of SARS-CoV-2‚ Influenza A‚ or Influenza B. Discard any expired kits responsibly. A valid expiration date ensures the reliability and effectiveness of the test‚ providing confidence in the outcome. Prioritizing this step is fundamental for obtaining trustworthy results.
Gathering Necessary Materials
Before beginning the 2San test‚ ensure you have all required components readily available to facilitate a smooth and accurate testing process. This includes the test device itself‚ the sterile nasal swab provided within the kit‚ and the extraction buffer tube with its accompanying dropper cap. A timer or stopwatch is also essential for precise result reading.
Additionally‚ prepare a clean‚ flat surface to work on‚ and have a biohazard disposal bag handy for safe disposal of used materials. Hand sanitizer is recommended for proper hygiene before and after the test. Avoid using any components from different kits‚ as they may not be compatible. Having everything organized beforehand minimizes errors and ensures a reliable testing experience.
Step-by-Step Testing Procedure
Begin with gentle nasal swab collection‚ followed by extracting the sample with the provided buffer. Carefully apply the extracted liquid to the test device‚ and await results.
Nasal Swab Collection Technique
Proper nasal swab collection is crucial for accurate test results. Begin by gently opening the sterile swab package‚ avoiding contact with the swab tip. Tilt your head slightly back. Carefully insert the entire swab tip into one nostril‚ approximately 1-2 centimeters (0.4-0.8 inches) – this should feel comfortable‚ not painful.
Slowly rotate the swab against the inside wall of your nostril at least five times‚ ensuring you collect an adequate sample. Repeat this process in the other nostril using the same swab. This dual-nostril collection maximizes sample acquisition. Remove the swab carefully‚ avoiding touching the swab tip to any surface.
Immediately proceed to the next step – extracting the sample – to prevent contamination and ensure the integrity of the collected material. A well-executed swab collection significantly impacts the reliability of your 2San test.
Extracting the Sample
Begin the extraction process by firmly inserting the nasal swab into the extraction tube containing the extraction buffer. Ensure the swab is fully submerged within the liquid. Rotate the swab vigorously within the buffer for at least 15 seconds‚ pressing the swab against the sides of the tube to release the collected sample.
While rotating‚ apply firm pressure to squeeze and extract as much liquid as possible from the swab. After rotation‚ pinch the sides of the tube to express any remaining liquid from the swab. Discard the swab by disposing of it properly – it is considered biohazardous waste.
Securely attach the provided dropper cap to the extraction tube. Gently mix the extracted sample by inverting the tube several times. This ensures a homogenous solution for accurate application to the test device. Proceed immediately to applying the sample.
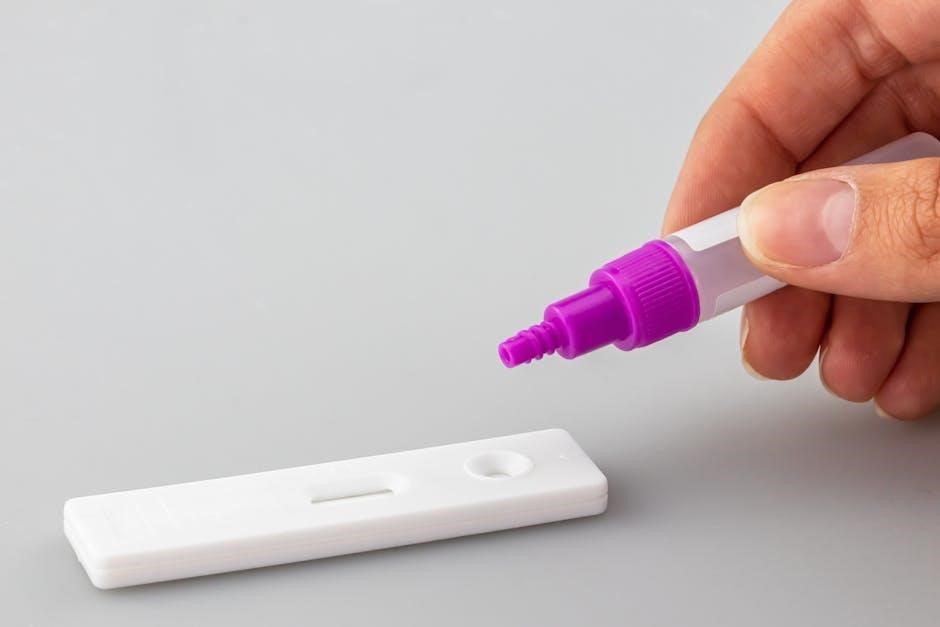
Applying the Sample to the Test Device
Hold the test device horizontally and carefully dispense exactly three drops of the extracted sample into the sample well (marked “S”) on the test cassette. Avoid creating air bubbles‚ as this can affect the test results. Ensure the drops fall directly into the well and do not overflow.
Immediately start the timer for the designated reading time – typically 15 to 30 minutes‚ as specified in the instructions for use. Do not move or disturb the test device during this incubation period. Any movement could compromise the accuracy of the test.
Observe the test results within the specified time frame. Results appearing after the reading time are considered invalid and should not be interpreted. Proper sample application is crucial for reliable detection of SARS-CoV-2 and influenza antigens.
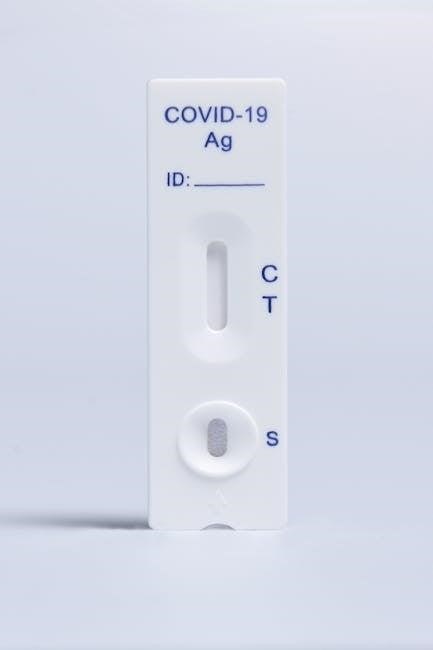
Interpreting Test Results
Positive results indicate the presence of antigens‚ while negative results suggest their absence. Invalid tests require repeating the procedure with a new kit.
Understanding Positive Results
A positive result on the 2San test means that antigens for SARS-CoV-2‚ Influenza A‚ or Influenza B were detected in your nasal sample. This suggests you likely have an active infection with one or more of these viruses. It’s crucial to follow current public health guidelines regarding isolation and symptom management.
A positive result doesn’t necessarily indicate the severity of the illness‚ but it does confirm infection. Consider consulting a healthcare provider for further evaluation‚ especially if you have underlying health conditions or experience severe symptoms. They can advise on appropriate treatment options and monitor your condition. Remember to inform your healthcare provider about your positive self-test result.
Even with a positive result‚ continue practicing preventative measures like masking and hand hygiene to protect others from potential spread.
Understanding Negative Results
A negative result from the 2San test indicates that antigens for SARS-CoV-2‚ Influenza A‚ and Influenza B were not detected in your nasal sample at the time of testing. However‚ a negative result does not guarantee you are free from infection. It’s possible you have a low viral load‚ are testing too early in the infection‚ or the sample collection wasn’t optimal;
If you continue to experience symptoms consistent with COVID-19 or influenza‚ even with a negative result‚ consider retesting in 1-2 days‚ especially if symptoms worsen. Continue to practice preventative measures like masking and social distancing to avoid potential spread.
Consult a healthcare provider if symptoms persist or worsen‚ as they can assess your condition and recommend further testing or treatment.
Invalid Test Results and Troubleshooting
An invalid test result is indicated when the control line (C) does not appear. This signifies the test didn’t function correctly‚ and the result is unreliable. Several factors can cause this‚ including insufficient sample volume‚ incorrect procedure followed‚ or a faulty test device.
Troubleshooting: Ensure you collected an adequate nasal swab sample and followed the extraction buffer instructions precisely. Verify the test device wasn’t damaged and is within its expiration date. If the control line still doesn’t appear after repeating the test with a new device‚ do not interpret the result.
Contact the manufacturer or your healthcare provider for assistance. A new test should be performed to obtain a valid result‚ and medical advice should be sought if symptoms persist.
Accuracy and Limitations of the Test
2San’s test demonstrates high sensitivity and specificity‚ but false positives or negatives can occur. Accuracy depends on proper sample collection and timing of testing.
Sensitivity and Specificity
Sensitivity refers to the test’s ability to correctly identify positive cases; a highly sensitive test minimizes false negatives. Specificity indicates the test’s ability to correctly identify negative cases‚ reducing false positives. The 2San test aims for high levels of both‚ but it’s crucial to understand these aren’t absolute.
Factors influencing these metrics include viral load‚ the timing of the test relative to symptom onset‚ and the accuracy of the nasal swab collection technique. Lower viral loads‚ especially early or late in infection‚ may lead to false negatives. Conversely‚ cross-reactivity with other viruses could potentially cause a false positive‚ though this is less common.
While the 2San test provides a valuable screening tool‚ it should not be the sole basis for clinical decisions. Confirmatory testing with a molecular assay (like PCR) may be necessary‚ particularly for negative results when clinical suspicion remains high.
Potential for False Positives/Negatives
False negative results can occur if the viral load is low‚ particularly early in infection or if the sample was collected improperly. Insufficient nasal swab depth or timing – testing too soon or too late after symptom onset – can impact accuracy. Repeat testing‚ as recommended in the instructions‚ can mitigate this risk.
False positive results are less frequent but possible. Cross-reactivity with other respiratory viruses or non-specific binding can occasionally lead to an incorrect positive reading. It’s important to consider your overall clinical picture and exposure history when interpreting results.
If symptoms persist despite a negative result‚ or if you have known exposure‚ consult a healthcare professional for further evaluation and potential confirmatory testing‚ such as a PCR test. The 2San test is a screening tool‚ not a definitive diagnosis.
Reporting Test Results
Report positive results to public health authorities and your healthcare provider promptly. Sharing data aids surveillance efforts and informs community health strategies effectively.
Public Health Reporting Requirements
Reporting positive 2San test results to public health departments is crucial for effective disease surveillance. This data contributes to understanding viral spread‚ identifying outbreaks‚ and informing public health interventions. Specific reporting requirements vary by location‚ so consult your local health authority’s guidelines.
Generally‚ reporting involves submitting patient information (while maintaining privacy) and test results through designated channels‚ often online portals or phone lines. Timely reporting allows for rapid response measures‚ such as contact tracing and resource allocation. Accurate data is vital for tracking trends and monitoring the effectiveness of control strategies. Failure to report may be subject to penalties‚ depending on local regulations.
Always adhere to your state or local health department’s specific instructions for reporting COVID-19 and Influenza test results.
Communicating Results to Healthcare Providers
Sharing your 2San test results with a healthcare provider is highly recommended‚ regardless of whether the result is positive or negative. This allows for appropriate medical guidance and treatment decisions. Your doctor can interpret the results in the context of your overall health and symptoms.
For positive results‚ a healthcare provider can advise on isolation protocols‚ symptom management‚ and potential antiviral therapies. Even negative results should be discussed‚ as they don’t always rule out infection‚ especially if symptoms persist. A healthcare professional can determine if further testing or evaluation is needed.
Keep a record of your test result and share it during telehealth appointments or in-person visits. Proactive communication ensures you receive the best possible care.

Respiratory Virus Surveillance Initiatives
Sentinel providers submit clinical samples to PHEL‚ aiding in tracking COVID-19‚ Influenza‚ and other respiratory illnesses for public health monitoring and response.
Role of Sentinel Providers
Sentinel providers play a crucial role in respiratory virus surveillance‚ acting as key data collection points within the broader public health network. These healthcare professionals‚ strategically located‚ routinely collect clinical samples from patients exhibiting symptoms indicative of respiratory infections – including COVID-19‚ Influenza A‚ Influenza B‚ and other related illnesses.
Specifically‚ they submit samples from individuals who have tested positive using tests like the 2San test‚ or those presenting with influenza-like illness. This submitted material undergoes advanced testing at PHEL (Public Health England Laboratory)‚ utilizing specialized surveillance algorithms. This process allows for detailed analysis‚ tracking viral prevalence‚ identifying emerging subtypes‚ and monitoring for genetic mutations – all vital for informed public health strategies and interventions.
Sample Submission Instructions to PHEL
PHEL (Public Health England Laboratory) requires specific protocols for submitting samples collected from patients suspected of having respiratory viral infections‚ including those identified through the 2San test. Detailed instructions‚ crucial for maintaining sample integrity and accurate surveillance data‚ are readily available online via the Respiratory Virus Surveillance Initiative Instructions for Laboratories document.
These guidelines cover proper specimen collection techniques‚ appropriate storage and transportation temperatures‚ required accompanying documentation – including patient demographics and clinical information – and designated shipping addresses. Adherence to these instructions is paramount to ensure reliable test results and effective monitoring of viral trends. Providers should consult the complete document for the most up-to-date procedures and any specific requirements related to COVID-19‚ Influenza A‚ or Influenza B.
Emergency Use Authorization (EUA) Information
2San’s COVID-19 and Influenza A/B test is authorized for emergency use only‚ requiring a prescription from a healthcare professional for legal use.
Understanding EUA Status
The 2San COVID-19 and Influenza A/B test has received Emergency Use Authorization (EUA) from relevant health authorities. This means the test has been authorized for use during the declared public health emergency‚ even though it hasn’t undergone the full‚ standard FDA review process.
The EUA allows for quicker availability of critical diagnostic tests during times of urgent need. However‚ it’s crucial to understand that EUA authorization is not the same as full FDA approval. The test’s performance characteristics continue to be monitored and evaluated.
This authorization is specifically for the detection of SARS-CoV-2‚ Influenza A‚ and Influenza B‚ and is intended for use by authorized laboratories and individuals with a valid prescription. The EUA status may change as more data becomes available or the public health emergency evolves.
Prescription Use Only
The 2San COVID-19 and Influenza A/B test is designated for prescription use only. This means a licensed healthcare professional must authorize the test before it can be legally obtained and used by an individual. This requirement ensures appropriate clinical context and guidance for test interpretation.
A healthcare provider can assess your symptoms‚ medical history‚ and potential exposure risks to determine if testing is necessary and appropriate. They can also provide counseling on what to do with the test results‚ regardless of whether they are positive or negative.
Obtaining a prescription ensures that the test is used responsibly and that individuals receive the necessary follow-up care if needed. Self-diagnosis and treatment based solely on home test results are discouraged; professional medical advice is essential.